Menu
- About
- For Research Patients
- Para Pacientes
- For Investigators
- For Industry Sponsors
close




A clinical trial is a type of research study that allows scientists to find better ways to prevent, diagnose, or treat certain diseases. They are the primary way that researchers learn whether a new test or treatment is safe and effective in people. Clinical trials can study many things, such as:
When found to be safe and helpful, the test, device or treatment is accepted by medical experts as the new standard of care.
Clinical trials are one of the last steps in a carefully designed research process that most often begins in a laboratory, where scientists first develop and test new ideas. Before any new treatment is used with people in clinical trials, researchers work for many years to understand its effects on cells in the laboratories and in animals.
The principal investigator is the person in charge of a clinical trial. He or she is an expert in what the clinical trial is about and is responsible for designing, managing, monitoring, and ensuring the integrity of a research project.
Clinical trials naturally advance through four phases of testing which normally includes finding the appropriate dosage, looking for side effects; evaluating therapeutic effectiveness; and post-approval surveillance to ensure that the drug is still safe for use in humans. The FDA will approve the drug or treatment after researchers ensure it is safe and effective during Phase I, II, and III trials.

https://www.ildcollaborative.org/resources/phase-iv-ipf-clinical-trials/
More than 90% of the population in the Rio Grande Valley is of Hispanic origin. However, less than 1% of participants of Hispanic origin are involved in clinical trials conducted in the United States.
It is therefore imperative that we make a concerted effort to engage and involve more people of Hispanic origin in innovative clinical trials that would not only benefit them but would also assist us in developing treatment strategies that are most effective for a predominately Hispanic population.
Below is an abbreviated list of additional benefits a patient can avail by being involved in clinical research.
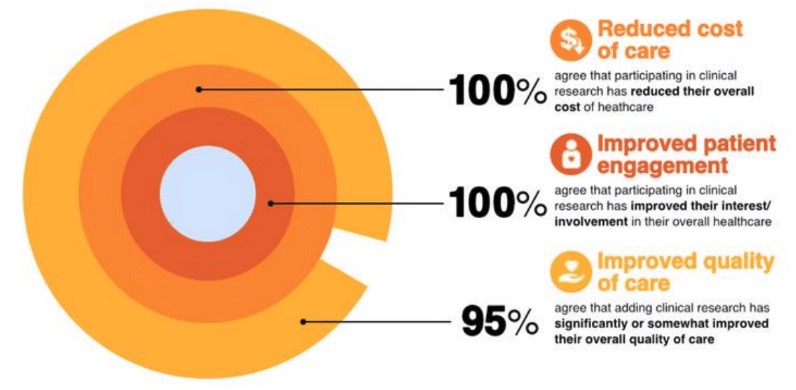
Figure 1. Benefits of clinical research participation. Used with permission. Quintiles: Clinical research participation as a care option. 2015 [Whitepaper]
DHR Health Institute for Research and Development is committed to bringing new treatments and cures to patients. Take a look at how our commitment to innovation supports our community!
Having an idea of what you can expect if you take part in a clinical trial can help relieve some of your concerns. Here's an overview of what might happen.
INFORMED CONSENT PROCESS
First, you will need to give your permission to take part in the study or clinical trial. Before any tests can be done to see if you are eligible for the clinical trial, you will need to give your written consent that you want to be a participant in the study.
A clinical coordinator or member of the research team will explain the study to you and your legal representative. Family members or friends may also be present, if you choose. They will make sure your questions and concerns are answered during this time.
You will be given an Informed Consent Form that outlines the key points of the study. Each Informed Consent Form should include the following:
If you decide to participate, you will need to sign the Informed Consent Form to move on to the next step. If you are undecided, you may take the Informed Consent Form home with you to review and discuss with family or friends. When you decide to participate, you may contact the study personnel to enroll in the study.
PARTICIPATING IN THE STUDY
Once you've signed the consent form, you'll be ready to participate in the study.
It is important to note that each study is different. Depending on the study, visits may be as short as a one-time blood draw or may take the course of several months. Each study will require different tests and procedures and these should be explained to you during the Informed Consent process.
For some studies, blood tests and/or imaging tests will be done to determine if you are eligible to participate and before you start any treatments. A medical history, physical exam, questionnaires or surveys may also be conducted.
You will most likely get more medical attention as a study participant than you would otherwise. Physicians and research staff may examine you more often and will want to know if you're having any side effects while being treated.
Clinical studies can take place in many locations, including the main hospital, doctors' offices, and the DHR Health Institute for Research and Development Clinical Research Unit. We have several clinical research coordinators and phlebotomists that are ready to assist with follow-up visits. A typical follow-up visit takes about 15 minutes, on average.
STUDY WITHDRAWAL
Participating in a study is voluntary. You can decide, at any point, for any reason, to withdraw from a study.
Your decision to withdraw or not participate will not affect the quality of your medical care and will not involve any penalty or loss of benefits to which you are otherwise entitled.
HOW TO PARTICIPATE
The team from the DHR Health Institute for Research and Development are committed to bringing innovative clinical trials and studies to our patients and our community. For information on the available clinical studies currently active, please click here.
Qualifications to successfully enroll in a clinical trial are provided in the protocol of each study. These rules are called “Inclusion and Exclusion Criteria” and are used to ensure all research sites have patients with similar health conditions. With the inclusion and exclusion criteria, researchers can properly compare the right patients for the trial and effectively study the overall effects of the treatments and/or procedures.
Once you find the clinical trial you are interested in using the national data base at https://clinicaltrials.gov/ , you can contact us for a quick over-the-phone prescreening. If you qualify the prescreening, we will schedule your appointment to one of our physicians and see if you meet the complete inclusion and exclusion criteria.
You may also qualify by participating in one of our many healthcare campaigns. These campaigns may provide you with basic information about your disease. With this information, our medical staff can assist you with selecting the most appropriate clinical trial or you may take the information to your personal doctor to discuss which research options are good for you.
Protection of patients in clinical trials is the paramount responsibility of the institution in which the study is being conducted. Federal, State, and institutional policies and procedure govern the conduct of clinical research. All Institutions involved in clinical research are required to provide assurance to the federal government that research will be conducted with:
The duration of a clinical trial is extremely variable. Depending on the complexity of the disease, a trial may be as short as a few months or as long as a few years. The goal is to allow optimal number of patients to participate in a clinical trial and monitor for adequate duration to validate the outcomes. You can visit https://clinicaltrials.gov/ for more information about the duration of active clinical trials or you may also contact us for more information on a clinical trial.
Yes! If in a clinical trial a drug, device, or any other intervention is the direct cause of the observed side effect; you will receive prompt medical care at no cost to you or to your health insurance. This is one benefit of being involved in a clinical trial as a research participant. You will be able to access free innovative treatment, frequent monitoring visits, and complete peace of mind that if a complication is encountered, you will be treated free-of-charge.
Before participating in a clinical trial, it is important that you understand how the study will be conducted and what direct and/or indirect benefit you will gain from this study. The examples below may be useful for any patient to ask before participating that will help you better understand the risks and benefits the study.
There may be many benefits to you and your community when you choose to participate in a clinical trial. If you choose to participate you can help doctors and researchers understand and answer challenging questions to improve healthcare for yourself and others struggling with similar health conditions. Some of these benefits include:
BENEFITS
RISKS
Before a drug, device, or any other intervention is tested on patients; testing is done in laboratory with animals and healthy volunteers to ascertain the safety and efficacy of the clinical trial. However, as is true for standard of care, any intervention carries with it are inherited risks which are listed below:
A protocol is a plan that clinical trials use in order to ensure the following:
A protocol ensures basic standards of care and research measures are the same across each research site.
The word “investigational” implies that the drug, device, diagnostic procedure, or an innovative surgical intervention has not yet been approved by the FDA for use in humans as a standard of care. This however, does not imply that it cannot be used in humans; it simply suggests that rigorous testing has to be done before it could be approved for wide spread use in humans as a standard of care for the treatment of intended diseases.
Before agreeing to participate in a clinical trial, make sure you understand the benefits and the risks in order to make informed decisions.
Most of the information will be given to you in a form of a document called “The Informed Consent”. This document is reviewed by an independent Institutional Review Board (IRB) and details all possible risks that have been reported or that may happen to you while in the trial. It will also tell you about the treatment you will receive, how it is believed to work, what will happen at each visit, and what to do in case you experience a side effect while in the clinical trial. This document will be given to you in a language you are most comfortable in comprehending. If you find it difficult to read or understand, the physician or a mentor of his/her team will be able to go over it with you.
The informed consent must be fully understood and signed by you before any research procedure can be performed. If at any point you do not agree with the informed consent or simply do not wish to continue you may do so without questions asked jeopardizing your medical care.
Clinical trials are conducted monthly in hospitals and clinics of physicians. It usually requires a dedicated team on investigators, coordinators, data integrators, etc. Any institution undertaking in these activities has to provide assurance to the Federal Department of Health and Human Services using an application form called Federal Wide Assurance for the Protection of Human Subjects (FWA). Under the U.S. Federal Policy, each institution that engages in clinical research has to ensure that, in the discharge of its responsibility, it will abridge by the statement of principle outlined in the Common Rule.
DHR Health Institution for Research and Development has provided assurance to the federal agencies ensuring conduct of clinical research that will protect the rights of every participants.
Funding for clinical research is largely provided through two major sources:
The most dominated public form of funding for clinical research is through the National Institute of Health (NIH); although, many other federal and state agencies may also provide funding for clinical research.
Other key sources of funding for clinical research are provided among private entities, pharmaceutical companies, device manufacturers, and developers of diagnostic tools. Funding by foundations, private donors, and endorsements are also used to support clinical trials.
